Surround Medical wins FDA 510(k) for PORTRAY™
Katherine E. Pfaff
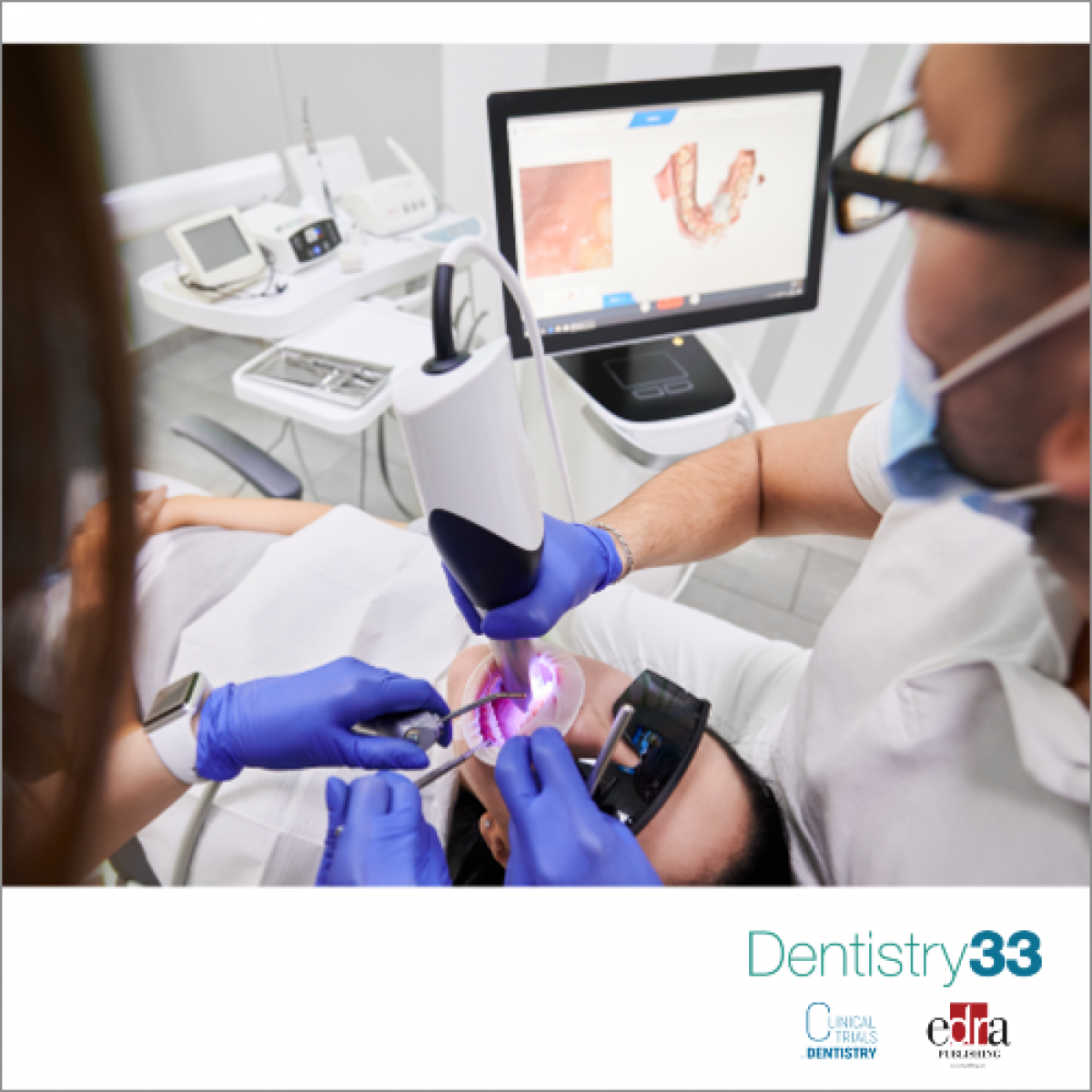
Surround Medical Systems, Inc. received FDA 510(k) pre-market clearance for the 2D diagnostic system, PORTRAY Dental Imaging System. The devices are expected to be available within the fourth quarter and will be prepared and shipped from the North Carolina facility.
Under the clearance, PORTRAY can be used as a 2D diagnostic tool, as well as to utilize additional diagnostic capabilities, including 2D synthetic and 3D tomosynthesis imaging to create radiographs of the jaw, teeth, and mouth. The advancement could improve cavity detection compared to x-rays by providing imaging of early tooth decay and improved detail to see additional dental issues.
"Dentists have been waiting for a major improvement to intraoral dental imaging for more than two decades," said David LaVance, president and CEO. "The PORTRAY system provides dentists with an intraoral 3D dental imaging system that will allow the dentists to provide a higher level of care for patients by detecting cavities and fractures that would otherwise go undetected by current 2D intraoral dental imaging systems. The PORTRAY system has the potential to substantially change and improve dental techniques and provide patients better outcomes and improved health."
PORTRAY was developed based on carbon nanotube technology that was built on x-ray methodology. The discoveries were made by the University of North Carolina at Chapel Hill researchers.
 Related articles
Related articles
News 11 May 2026
Food and Drug Administration (FDA) has granted FDA clearance for its Dx Software following a review completed on April 10, 2026.
Products 22 December 2025
AI dental company announces 510(k) clearance from the U.S. Food and Drug Administration for Overjet CBCT Assist.
News 05 November 2025
Perimetrics, Inc., a dental technology company pioneering quantitative diagnostics, announced today that the U.S. Food and Drug Administration (FDA) has granted clearance for the InnerView...
Products 26 November 2024
DentalMonitoring Enhances Orthodontic Software With FDA-Cleared AI Technology
DentalMonitoring, the leader in artificial intelligence (AI)-powered remote monitoring for orthodontics, has released a major software update introducing features and indications based on the...
News 15 October 2024
Relu Secures FDA Clearance and CE Mark Approval for Revolutionary Dental AI Tool
Relu, a pioneer in artificial intelligence (AI)-assisted segmentation for dental labs and software companies, proudly announces the dual achievement of 510(k) clearance by the U.S. Food and Drug...
 Read more
Read more
New initiative invites dentists to experience DEXIS’ most advanced AI yet, built on scale, speed, and clinical trust.
News 05 June 2026
(Nasdaq: ALGN), a leading global medical device company that designs, manufactures, and sells the Invisalign® System of clear aligners, iTero™ intraoral scanners, and exocad™ CAD/C
News 05 June 2026
(Nasdaq: HSIC), the world’s largest provider of health care solutions to office-based dental and medical practitioners, today announced that its Board of Directors has elected Will
As the University of Colorado School of Dental Medicine celebrates the graduating DDS Class of 2025, we are proud to recognize the students and faculty members whose exceptional de
Oral surgery 05 June 2026
This peer-reviewed oral surgery article summarizes clinical evidence from International journal of oral and maxillofacial surgery (2026). It focuses on findings that may help dental professionals...