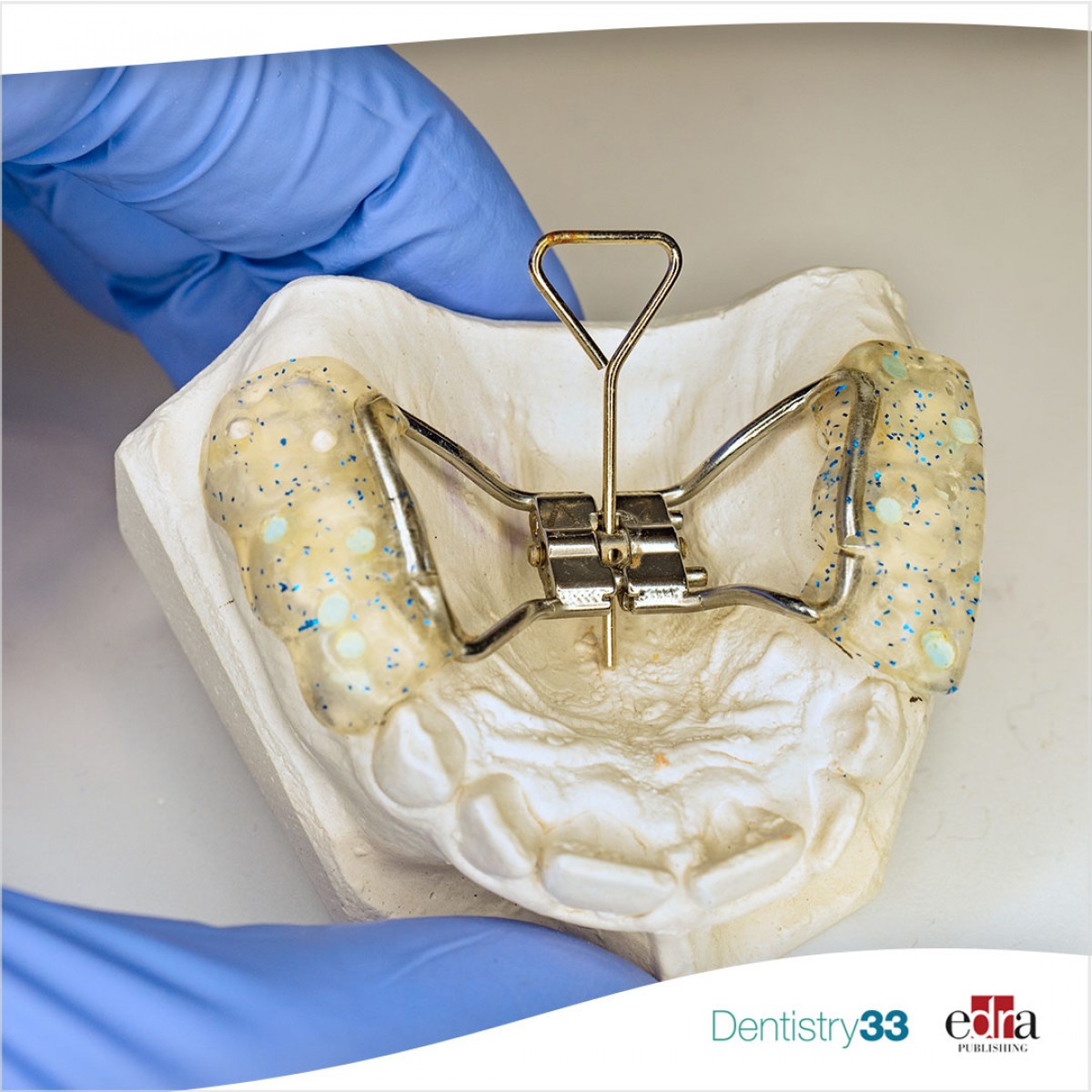
ADA asking public, profession to report concerns with certain palatal expanders
By David Burger, ADA News
The American Dental Association (ADA) is encouraging the public and dental profession to follow the Federal Food & Drug Administration’s process of promptly reporting adverse events or serious complications related to certain dental devices that are fixed (nonremovable) palatal expanders.
According to an April 3 ADA News story, the palatal expanders of concern were named in a March 30 FDA announcement and include:
- Anterior growth guidance appliance and fixed anterior growth guidance appliance.
- Anterior remodeling appliance and fixed anterior remodeling appliance.
- Osseo-restoration appliance and fixed osseo-restoration appliance.
- Any other similar device types.
“The FDA is aware of these devices being used to treat conditions such as obstructive sleep apnea and temporomandibular joint disorder of the jaw, and to remodel the jaw in adults,” according to the announcement. “However, the safety and effectiveness of these devices intended for these uses have not been established, and these devices are not cleared or approved by the FDA.”
The FDA asks patients, caregivers and health care providers to report any complications with those named devices to the FDA.
The ADA is encouraging the public and dental profession to report concerns to MedWatch to ensure any pertinent information related to these devices can be thoroughly evaluated by the FDA with regard to safety and efficacy. The ADA will continue to monitor for FDA updates regarding these devices.
If someone experiences any issues with any medical device, the FDA encourages them to file a voluntary report through MedWatch or call 800-332-1088 for more information on how to mail or fax the form.
Health care personnel employed by facilities that are subject to the FDA's user facility reporting requirements should follow the reporting procedures established by their facilities.
According to the announcement, the FDA is not aware of safety concerns related to orthodontic use of palatal expanders in children and adolescents. The safety and effectiveness of fixed (nonremovable) palatal expanders being used to treat conditions such as OSA and TMD, or to remodel the jaw in adults, have not been established and these devices have not been cleared or approved by the FDA.
For questions, email the Division of Industry and Consumer Education at DICE@fda.hhs.gov or call 800-638-2041 or 301-796-7100.
 Related articles
Related articles
News 26 May 2025
The Invisalign Palatal Expander is applicable to skeletal and dental malocclusion in primary, mixed, and permanent dentition and will be commercially available during the second half of 2025.
News 21 December 2023
Align Technology Receives US FDA 510(k) Clearance for the Invisalign Palatal Expander System
Previously introduced at the Align Technology Investor Day in September 2023, the Invisalign Palatal Expander System is a modern and innovative direct 3D printed device based on proprietary and...
Market 19 December 2023
Health Canada Updates License for Invisalign Palatal Expander System Expansion
Align Technology has announced that Health Canada issued an updated medical device license to Align for its Invisalign Palatal Expander System*. The updated license is for broad patient...
Digital Dentistry 23 February 2021
RAPID PALATAL EXPANDER: MAXILLARY AND MANDIBULAR MEASUREMENTS USING MANUAL AND DIGITAL METHODS
Ileana Carminati, Joanna Katarzyna Nowakowska, Anna D’Apote, Massimo Del Fabbro, Luca Francetti
Objectives: The aim of this work is to establish a correlation between rapid palatal expansion and changes in width, length and perimeter of the upper and lower arch.Materials and methods: The study...
Orthodontics 04 June 2026
This peer-reviewed orthodontics article summarizes clinical evidence from PloS one (2026). It focuses on findings that may help dental professionals evaluate treatment decisions, patient outcomes, or...
 Read more
Read more
USA 07 June 2026 - 30 November -0001
ICOI World Congress USA 2026: Event Preview and Professional Highlights
The ICOI World Congress USA 2026 will take place from August 13, 2026 to August 15, 2026 in TBD, USA, offering dental professionals a focused environment for continuing education, clinical updates...
Edra Professional Books 06 June 2026
Understanding Dental Insurance: A Practical Reference for Dental Professionals
Understanding Dental Insurance is an Edra professional dentistry reference focused on clinical practice, education and treatment planning.
New initiative invites dentists to experience DEXIS’ most advanced AI yet, built on scale, speed, and clinical trust.
News 05 June 2026
(Nasdaq: ALGN), a leading global medical device company that designs, manufactures, and sells the Invisalign® System of clear aligners, iTero™ intraoral scanners, and exocad™ CAD/C
News 05 June 2026
(Nasdaq: HSIC), the world’s largest provider of health care solutions to office-based dental and medical practitioners, today announced that its Board of Directors has elected Will