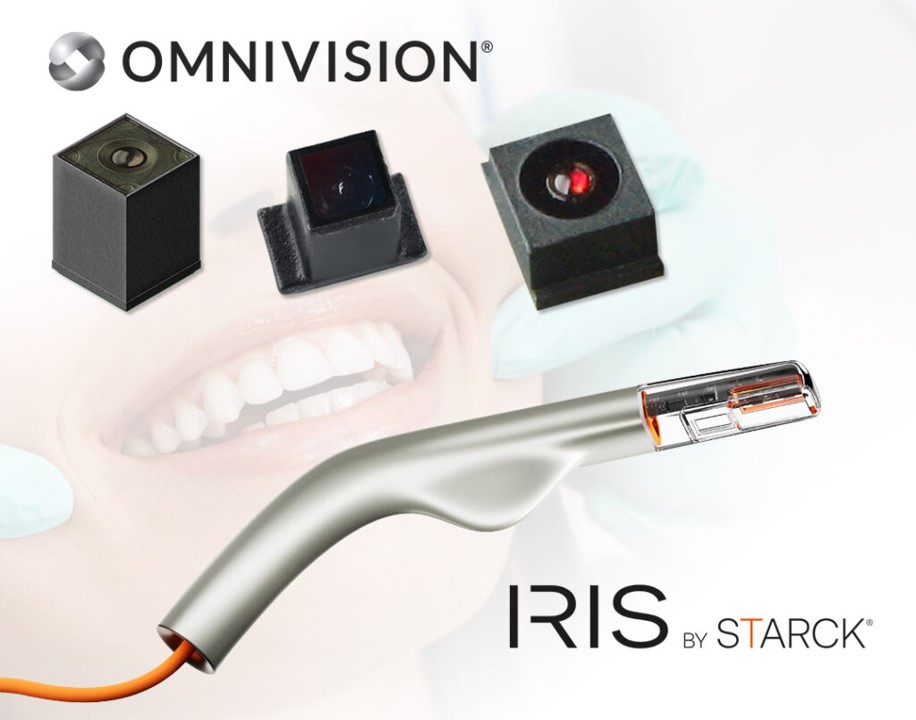
OMNIVISION Powers Biotech’s Intraoral Scanner
OMNIVISION, a leading global developer of semiconductor technology—including advanced digital imaging, analog, and display solutions—and Biotech Dental, a specialist in the design and manufacture of medical devices and digital solutions for dental applications, today announced a partnership involving Biotech Dental’s use of multiple medical-grade camera modules from OMNIVISION in the design of its new Scan4All 3D intraoral scanners, Iris by Starck. The camera modules for dental applications feature OMNIVISION’s CameraCubeChip® technology with an integrated lens and will be showcased at the International Dental Show (IDS), March 25-29, 2025, in Cologne, Germany (Hall 4.2/Booth #H038).
The Scan4All intraoral scanner, Iris by Starck, is a handheld device slightly larger than an electric toothbrush. It uses sophisticated software to develop a 3D digital model of a patient’s teeth and gums, which can then be used to create dental restorations and implants. This method is significantly more convenient and accurate than the traditional dental putty approach. However, most intraoral scanners available today have only one image sensor, limiting their speed and accuracy. Biotech Dental is addressing this pain point by partnering with OMNIVISION to develop intraoral scanners with multiple image sensors that produce quick and accurate high-quality 3D dental models.
“We partnered with OMNIVISION due to the wide range of imaging solutions they offer for dental applications. Most importantly, OMNIVISION’s camera modules are developed and tested specifically for the medical and dental markets and have a long lifetime, which, in turn, ensures that our intraoral scanners retain their value and remain a high-quality solution for our customers,” said Philippe Veran, CEO of Biotech Dental.
According to Ehsan Ayar, medical product marketing manager at OMNIVISION, “Biotech Dental offers innovative, high-quality solutions for the dental industry. We are excited to partner with them to enhance their next-generation intraoral scanners, which will use several of our medical-grade high-resolution image sensors, taking dental visualization to the next level. We are the only company that offers a wide range of medical-grade imagers, which go through extensive biocompatibility, sterilization, and medical tests. This gives dental professionals peace of mind during procedures.”
3D intraoral scanners are quickly replacing traditional dental putty impressions, which are time-consuming, complex, and can be inaccurate. Data Bridge Market Research expects the global dental intraoral scanners market to grow at a compound annual growth rate (CAGR) of 11.1% from 2023 to 2030 and to reach USD 1,325.80 million by 2030, up from USD 573.76 million in 2022.¹
Biotech Dental will utilize the following OMNIVISION CameraCubeChip® camera modules:
The OCH2B30 camera module utilizes a square-format CMOS image sensor with a 1500 x 1500 resolution at 60 frames per second (fps) or 720p resolution at 120 fps.
The OCHSA10 camera module offers 800 x 800 resolution with a frame rate of 60 fps or 400 x 400 resolution at 90 fps for blur-free images and video.
The OVM9724 ultra-compact front-facing camera module captures 720p high-definition video at 30 fps.
Both the OCH2B30 and OCHSA10 utilize OMNIVISION’s PureCel®Plus-S stacked-die technology, which enables their compact size. The OVM9724 is built on OMNIVISION’s powerful OmniBSI™+ pixel architecture to enable high-quality color images and fast-frame video. CameraCubeChip® is a reflowable, all-in-one camera drop-in solution—eliminating the need for additional optical components, which significantly streamlines manufacturing. The combination of excellent image quality and a small form factor makes these camera modules ideal for integration into slim, compact devices, such as the tip of a 3D intraoral scanner.
OMNIVISION is ISO 13485-certified; its camera modules are tested for electromagnetic interference (EMI) and electromagnetic compatibility (EMC) compliance and sterilization. They are also biocompatible and waterproof. Due to OMNIVISION’s rigorous production testing, they are guaranteed to meet the stated electrical, mechanical, and optical specifications and do not require tuning or calibration.
About OMNIVISION
OMNIVISION is a global, fabless semiconductor organization that develops advanced digital imaging, analog, and display solutions for multiple applications and industries, including mobile phones, security and surveillance, automotive, computing, medical, machine vision, and emerging applications. Its award-winning, innovative technologies enable a smoother human/machine interface in many of today’s commercial devices.
Source: www.ovt.com
 Related articles
Related articles
Dentists Choice announces it insured 8,000 dentists in its first 150 days by leveraging Counterpart’s Agentic Insurance™ platform.
News 30 May 2025
Pearl Powers AI Transformation Across Partnerships for Dentists’ Expansive Practice Network
Pearl, the global leader in dental AI solutions, and Partnerships for Dentists (“P4D”), a leading dental support organization serving practices across the United States, today announced some of...
Products 18 September 2024
S.M.I.L.E. Healthcare Pathway Program Empowers Diverse Youth to Pursue Careers in Dentistry
Henry Schein, Inc. (Nasdaq: HSIC), the National Dental Association (NDA), and the Schattner Foundation recently completed the second year of an innovative program designed to inspire students from...
Maeva Dental Advisors is pleased to announce the launch of Pronto! Dentistry’s Practice Performance System
Products 15 May 2024
Inspiring the Next Generation: Dr. Paul Carey Scholarship Empowers Future Medical Leaders
Dr. Paul Carey, DDS, a distinguished figure in the medical community, is proud to announce the continuation of the Dr. Paul Carey Scholarship for Medical Students, an esteemed scholarship program...
 Read more
Read more
Dentistry Today recently attended a robotic-assisted dental implant procedure performed by Jay Neugarten, DDS, MD, FACS, utilizing the Yomi S robotic guidance s
Endodontics 21 April 2026
For a daily dental practice, the Panoramic (PANO) X-ray film is one of the most commonly used dental X-rays.
Editorials 21 April 2026
NYU Dentistry Receives $5M to Expand Oral Health Care for Children with Disabilities
State funding will create dedicated space for pediatric and adolescent care and improve access for people with disabilities across the lifespan